
#157 TMA
3,4,5-TRIMETHOXYAMPHETAMINE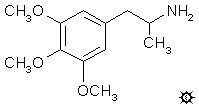
|
| [3D .mol structure] |
Under an inert atmosphere, 38 g LAH was wetted with 100 mL anhydrous Et2O, and then suspended in 1 L dry THF. This was brought up to a gentle reflux, and there was added, slowly, a solution of 43.7 g 2-nitro-1-(3,4,5-trimethoxyphenyl)propene in 160 mL THF. Refluxing was continued for 36 h, and then the reaction mixture was cooled with an external ice bath. The excess hydride was destroyed by the cautious addition of 38 mL H2O, and this was followed by 38 mL 15% NaOH, and finally another 114 mL H2O. The inorganic salts which should have ended up as a loose, granular, easily filterable mass, looked rather like library paste, but they were filtered nonetheless. Washing with THF was attempted, but it was not efficient. The combined filtrate and washes were stripped of solvent under vacuum giving 31.5 g of the crude base as an amber oil. This was dissolved in 140 mL IPA, neutralized with concentrated HCl (15 mL was required), and diluted with 650 mL anhydrous Et2O. There was an initial oily phase which on continued stirring changed to pale pink solids. These were finely ground under CH3CN to give 15.2 g of 3,4,5-trimethoxyamphetamine hydrochloride (TMA) as white crystals that melted at 195-211 °C. All aluminum salts from everywhere were dissolved in dilute HCl, and 1 Kg of potassium sodium tartrate was added. There as added 25% NaOH allowed the pH to bring the pH to >9 without the precipitation of basic alumina. Extraction of this phase with CH2Cl2 was followed by removal of the solvent and salt formation as described above, allowed the isolation of an additional 6.4 g TMA. The product prepared in this manner contains some 10-15% 3,5-dimethoxy-4-hydroxyamphetamine as an impurity. A solution of 20 g of the TMA made in this manner in 200 mL 5% NaOH was extracted with 2x200 mL CH2Cl2. The pooled extracts were washed with 4x100 mL 5% NaOH, and the aqueous washes were pooled with the original base phase. The organic phase was stripped of its CH2Cl2 under vacuum to give an oil that was dissolved in 40 mL IPA, neutralized with concentrated HCl, and diluted with 400 mL anhydrous Et2O. There was the immediate formation of spectacular white crystals of pure 3,4,5-trimethoxyamphetamine hydrochloride, weighing 15.4 g and having a mp of 220-221 °C. The aqueous phase was brought to neutrality, treated with 10 g potassium di-hydrogen phosphate, brought to pH 9.0 with the careful addition of NaOH, and extracted with 5x100 mL CH2Cl2. Evaporation of the solvent under vacuum gave an oil that spontaneously crystallized. This product, 3,5-dimethoxy-4-hydroxyamphetamine could be further purified by sublimation at 130 °C at 0.2 mm/Hg. It was a white crystalline solid that slowly discolored in the air. The literature describes a picrate salt with a mp of 225 °C from EtOH.
DOSAGE: 100 - 250 mg.
DURATION: 6 - 8 h.
QUALITATIVE COMMENTS: (with 135 mg) I had no nausea, although I always vomit with mescaline. Somehow my personality was divided and exposed, and this allowed me to understand my psychic structure more clearly. But maybe others could look in there, too. The psychiatric use of this drug would be interesting to pursue. It is not completely pleasant, maybe because of this personal intimacy.
(with 140 mg) There were not the color changes of mescaline there, but certainly a good humor and an over-appreciation of jokes. The images behind the eyes were remarkable and tied in with the music, and I became annoyed at other people's conversations that got in the way. I was out of it in eight hours. I would equate this to 300 or 350 milligrams of mescaline and I rather think that I would prefer the latter.
(with 225 mg) There was quite a bit of nausea in the first hour. Then I found myself becoming emotionally quite volatile, sometimes gentle and peaceful, sometimes irritable and pugnacious. It was a day to be connected in one way or another with music. I was reading Bernstein's 'Joy of Music' and every phrase was audible to me. On the radio, Rachmaninoff's 2nd piano concerto on the radio put me in an eyes-closed foetal position and I was totally involved with the structure of the music. I was suspended, inverted, held by fine filigreed strands of the music which had been woven from the arpeggios and knotted with the chords. The commercials that followed were irritating, and the next piece, Slaughter on Fifth Avenue, made me quite violent. I was told that I had a, 'Don't cross me if you know what is good for you,' look to me. I easily crushed a rose, although it had been a thing of beauty.
EXTENSIONS AND COMMENTARY: TMA was the very first totally synthetic psychedelic phenethylamine that was found to be active in man, for which there had been any attempt to describe such drug effects in any detail. This was the report of research done in Canada, and it appeared in 1955, six years before my own report on the material. There was an earlier report on TMPEA which is mentioned in the appropriate recipe, but there were few details given. Also there had been interest in reports that adrenalin that had become old and discolored seemed to elicit central effects in man. The oxidation products were identified as the deeply colored indolic compound adrenochrome and the colorless analogue adrenolutin. The controversy that these reports created just sort of died away, and the adrenochrome family has never been accepted as being psychedelic. No one in the scientific community today is looking in and about the area, and at present this is considered as an interesting historical footnote. But, in any case, they are not phenethylamines and so not part of this book.
The Canadian studies with TMA involved the use of a stroboscope as a tool for the induction of visual phenomena. These experiments used levels in the 50-150 milligram range, and generally employed pre-treatment with Dramamine for the successful prevention of nausea. There was reported giddiness and light-headedness, and some remarkable flash-induced visualizations. With higher levels, the visual syntheses are present without external stimulation. But there is a thread of negativity that seems to pervade the experience at these higher levels, and the appearance of a publication that emphasized the possible antisocial nature to TMA seemed to discourage further medical exploration. Military interest was maintained however, apparently, as TMA became a part of the chemical warfare studies where it was referred to with the code name EA-1319. It had been used in human trials with psychiatric patients, but no details of these experiments have been published.
The presence of a potentially active impurity in TMA deserves some comment. In the Canadian work, the material used was described as melting at 219-220 °C, which is the property given for the impurity-free material above. If this was the actual material used in those studies, this impurity (3,5-dimethoxy-4-hydroxyamphetamine) was probably not present. The Army studies use a material of unreported melting point. In my own studies, the lower melting product was used. There is an intriguing and unanswered question: what contribution did this phenolic component make to the nature of the observed effects of TMA? Assays on the isolated contaminant could answer that, but they have not yet been made.
There is an old saying that has gotten many people into trouble: "If one is good, then two is better." And if a statement of the measure of worth of a compound can be made from its potency, then TMA is a step in the right direction. And this was a chemically simple direction to follow further. Looking at mescaline as a compound with no carbons on its side-chain, and TMA as a mescaline molecule with one carbon on its side chain, then what about a compound with two carbons there, or three, or nine carbons?
Using this pattern of naming, TMA can be seen as alpha-methylmescaline, or AMM. And the two carbon homologue would be alpha-ethyl mescaline, or AEM. Its proper name is 2-amino-1-(3,4,5-trimethoxyphenyl)butane. It and its several higher homologues are discussed in a separate recipe entry called AEM (#1).
A final comment. But maybe a long one! Elsewhere, I have made comparisons between myristicin and MMDA, and between safrole and MDA. And here there is a similar parallel between elemicin and TMA. What are these relationships between the essential oils and the amphetamines? In a word, there are some ten essential oils that have a three carbon chain, and each lacks only a molecule of ammonia to become an amphetamine. So, maybe these essential oils, or "almost" amphetamines, can serve as an index for the corresponding real amphetamine counterparts. I had originally called this family the "natural" amphetamines, but my son suggested calling them the "essential" amphetamines, and I like that. At the time that I had synthesized TMA, back there in the '50s, I had the impulse to explore this body of Essential Amphetamines. As the old folk-wisdom says: "Nature is trying to tell us something."
One of the banes of the archivist is having to choose one pattern of organization over another. The book store owned by a language scholar will have the German poets and playwrights and novelists here, and the French ones over there. Next door, the book store is run by a letters scholar, and the poetry of the world is here, and the plays of the world are there, regardless of the language of origin. The same obtains with spices, and essential oils, and amphetamines. The spice cabinet is a rich source of chemical treasures, each source plant containing a host of com-pounds, some of which are true essential oils. And the next spice from the next plant has some of the same components and some new ones. Does one organize by plant (spice or herb) or by essential oil (amphetamine)? Let's do it by the ring substitution pattern of the amphetamine, and gather the spices and oils as a secondary collection.
(1) The 4-methoxy pattern. The pivotal essential oil is 4-allylanisole, or methyl chavicol, or estragole (called esdragol in the old literature). This allyl compound is found in turpentine, anise, fennel, bay, tarragon, and basil. Its smell is light, and reminiscent of fennel. The propenyl analogue is called anethole, or anise camphor, and it is found in both anise and camphor. It is a waxy solid, and has a very intense smell of anise or fennel. At low concentrations, it is sweet, as in magnolia blossoms, where it is also found. The drinks that turn cloudy with water dilution (Pernod-like liqueurs, and ouzo and roki), are heavy with it, since it was the natural flavoring in the original absinthe. That drink was very popular in the last century, as an intoxicant which produced an altered state of consciousness beyond that which could be ascribed to alcohol alone. It contained wormwood, which proved to be neurologically damaging. The flavorings, such as anethole, are still big things in synthetic liqueurs such as vermouth. Old anethole, when exposed to air and light, gets thick and sticky and yellowish, and becomes quite disagreeable to taste. Maybe it is polymerizing, or maybe oxidizing to stuff that dimerizes. Whatever. These changes are why old spices in the cabinet are best discarded. And adding ammonia to any of these natural product oils produces, in principle, 4-methoxyamphetamine, 4-MA.
(2) The 3,4-dimethoxy pattern. The main actor here is methyleugenol, or 4-allyl-1,2-dimethoxybenzene. This is located in almost every item in the spice cabinet. It is in citronella, bay (which is laurel, which is myrtle), pimiento, allspice, pepper, tree-tea oil, and on and on. It has a faint smell of cloves, and when dilute is immediately mistaken for carnations. The propenyl analogue is, not unreasonably, methylisoeugenol, a bit more scarce, and seems to always be that little minor peak in any essential oil analysis. The compounds missing that methyl group on the 4-oxygen are famous. The allyl material is eugenol, 4-allylguaiacol, and it is in cinnamon, nutmeg, cloves, sassafras and myrrh. You taste it and it burns. You smell it and think immediately of cloves. And its property as an anesthetic, in the form of a clove, is well known in the folk-treatment of toothaches. Actually, flowers of clove (the gillyflower, like the carnation) are the small, pointy things that decorate baked hams and, when stuck into apples, make pomander balls. This anesthetic property has recently led to a drug abuse fad, called clove cigarettes. Very strong, very flavorful, and very corrosive things from Southeast Asia. The eugenol that is present numbs the throat, and allows many strong cigarettes to be smoked without pain. The propenyl analogue is isoeugenol, with a smell that is subtle but very long lasting, used more in soaps and perfumes than in foods. The amine addition to the methyleugenol world produces 3,4-dimethoxyamphetamine, or 3,4-DMA. The isomer with the other methyl group missing is chavibetol (3-hydroxy-4-methoxyallylbenzene) and is found in the pepper leaf that is used with betel nut. A couple of positional rearrangement isomers of methyleugenol are known in the plant world. The 2,4-isomer is called osmorrhizole, and the conjugated form is isoosmorrhizole or nothosmyrnol; both are found in carrot-like vegetables. They, with ammonia, would give 2,4-DMA. And the 3,5-dimethoxyallylbenzene isomer from artemisia (a pungent herb commonly called mugwort) and from sage, would give rise to 3,5-DMA. This is an unexplored isomer which would be both an antidote for opium as well as a stimulant, if the classical reputation of mugwort is transferred to the amphetamine.
(3) The 3,4-methylenedioxy pattern. One of the most famous essential oils is safrole, or 4-allyl-1,2-methylenedioxybenzene. This is the mainstay of sassafras oil, and it and its conjugated isomer isosafrole have a smell that is immediately familiar: root beer! These are among the most widely distributed essential oils, being present in most of the spices, including the heavies such as cinnamon and nutmeg. I am not aware of the 2,3-isomer ever having been found in nature. Adding ammonia to either would give MDA.
(4) The 3-methoxy-4,5-methylenedioxy pattern. The parent compound is myristicin, 5-allyl-1-methoxy-2,3-methylenedioxybenzene, and the source of this is nutmeg (or the botanically parallel material, mace). The nutmeg is the seed of the tree Myristica fragrans and mace is the fibrous covering of the seed. The two spices are virtually identical as to their chemical composition. Myristicin and the conjugated isomer isomyristicin are also found in parsley oil, and in dill. This was the oil that was actually shown to be converted to MMDA by the addition of ammonia by passage through an in vitro liver preparation. So here is the major justification for the equation between the essential oils and the Essential Amphetamines. Care must be taken to make an exact distinction between myristicin (this essential oil) and myristin (the fat) which is really trimyristin or glyceryl trimyristate from nutmeg and coconut. This is the fat from myristic acid, the C-14 fatty acid, and these two similar names are often interchanged even in the scientific literature.
(5) The 2-methoxy-3,4-methylenedioxy pattern. This is the second of the three natural methoxy methylenedioxy orientations. Croweacin is 2-methoxy-3,4-methylenedioxyallylbenzene, and it takes its name from the binomial for the plant Eriostemon crowei from the worlds of rue and the citrus plants. It corresponds to the essential amphetamine MMDA-3a. This oil is found in plants of the Family Rutaceae. My memories of this area of botany are of Ruta graveolens, the common rue, whose small leaves smelled to me, for all the world, like cat urine. This plant has always fascinated me because of a most remarkable recipe that I was given by a very, very conservative fellow-club member, one evening, after rehearsal. He told me of a formula that had provided him with the most complete relief from arthritic pain he had ever known. It was a native decoction he had learned of many years eariler, when he was traveling in Mexico. One took equal quantities of three plants, Ruta graveolens (or our common rue), Rosmarinus officinalis (better known as rosemary), and Cannabis sativa (which is recognized in many households simply as marijuana). Three plants all known in folklore, rue as a symbol for repentance, rosemary as a symbol of remembrance, and pot, well, I guess it is a symbol of a lot of things to a lot of people. Anyway, equal quantities of these three plants are allowed to soak in a large quantity of rubbing alcohol for a few weeks. Then the alcoholic extracts are clarified, and allowed to evaporate in the open air to a thick sludge. This then was rubbed on the skin, where the arthritis was troublesome, and always rubbed in the direction of the extremity. It was not into, but onto the body that it was applied. All this from a very conservative Republican friend!
The methoxy-methylenedioxy pattern is also found in nature with the 2,4,5-orientation pattern. The allyl-2,4,5-isomer is called asaricin. It, and its propenyl-isomer, carpacin, are from the Carpano tree which grows in the Solomon Islands. All these plants are used in folk medicine. These two systems, the 2,3,4- and the 2,4,5-orientations, potentially give rise, with ammonia, to MMDA-3a and MMDA-2.
(6) The 3,4,5-trimethoxy pattern. Elemicin is the well studied essential oil, 5-allyl-1,2,3-trimethoxybenzene, primarily from the oil of elemi. It is, like myristicin, a component of the Oil of Nutmeg, but it is also found in several of the Oils of Camphor, and in the resin of the Pili in the Philippines. This tree is the source of the Oil of Elemi. I had found a trace component in nutmeg many years ago that proved to be 5-methoxyeugenol, or elemicin without the 4-methyl group; it is also present in the magnolia plant. The aldehyde that corresponds to this is syringaldehyde, and its prefix has been spun into many natural products. Any natural product with a syring somewhere in it has a hydroxy between two methoxys. The amphetamine base from elemicin or isoelemicin would be TMA, the topic of this very recipe.
(7) The 2,4,5-trimethoxy pattern. There is an essential oil called asarone that is 2,4,5-trimethoxy-1-propenylbenzene. It is the trans- or alpha-isomer, and the cis-isomer is known as beta-asarone. It is the isomerization analogue of the much more rare 1-allyl-2,4,5-trimethoxybenzene, gamma-asarone, or euasarone, or sekishone. Asarone is the major component of Oil of Calamus obtained from the rhizomes of Acorus calamus, the common Sweet Flag that grows wild on the edges of swamps throughout North America, Europe, and Asia. It has been used as a flavoring of liqueurs and, as almost every other plant known to man, has been used as a medicine. In fact, in Manitoba this plant was called Rat-root by the Cree Indians in the Lake Winnipeg area known as New Iceland, and Indian-root by the Icelandic pioneers. It was used externally for the treatment of wounds, and internally for most illnesses. There apparently is no report of central effects. The corresponding propanone, acoramone (or 2,4,5-trimethoxyphenylacetone), is also present in Oil of Calamus. The styrene that corresponds to asarone is found in a number of plants, and is surprisingly toxic to brine shrimp. The older literature describes an allyl-trimethoxy benzene called calamol, but it has never been pinned down as to structure. The isolation of gamma-asarone or euasarone from Oil of Xixin (from wild ginger) has given rise to a potential problem of nomenclature. One of the Genus names associated with wild ginger is Asiasarum which looks very much like the name asarone, which comes from the Genus Acorus. And a second Genus of medical plants also called wild ginger is simply called Asarum. There is an Asarum forbesi from central China, and it is known to give a pleasant smell to the body. And there is Asarum seiboldi which is largely from Korea and Manchuria. It has many medical uses, including the treatment of deafness, epilepsy, and rheumatism. The amphetamine that would arise from this natural treasure chest is TMA-2.
(8) The 2,5-dimethoxy-3,4-methylenedioxy pattern. The parent allyl benzene is apiole (with a final "e") or parsley camphor, and it is the major component of parsley seed oil. Its conjugated isomer is called isoapiole, and they are valuable as the chemical precurors to the amination product, DMMDA. Whereas both of these essential oils are white solids, there is a green oily liquid that had been broadly used years ago in medicine, called green, or liquid apiol (without the final "e"). It comes from the seeds of parsley by ether extraction, and when the chlorophyll has been removed, it is known as yellow apiol. With the fats removed by saponification and distillation, the old term for the medicine was apiolin. I would assume that any of these would give rise to white, crystalline apiole on careful distillation, but I have never tried to do it. The commercial Oil of Parsley is so readily available.
(9) The 2,3-dimethoxy-4,5-methylenedioxy pattern. The second of the three tetraoxygenated essential oils is 1-allyl-2,3-dimethoxy-4,5-methylenedioxybenzene, commonly called dillapiole and it comes, not surprisingly, from the oils of any of the several dill plants around the world. It is a thick, almost colorless liquid, but its isomerization product, isodillapiole, is a white crystalline product which melts sharply. This, by the theoretical addition of ammonia, gives DMMDA-2.
(10) The tetramethoxy pattern. The third and last of the tetra-oxygenated essential oils, is 1-allyl-2,3,4,5-tetramethoxybenzene. This is present as a minor component in the oil of parsley, but it is much more easily obtained by synthesis. It, and its iso-compound, and the amination product, are discussed under the last of theTen Essential Amphetamines, TA.
One must remember that the term "essential" has nothing to do with the meaning of needed, or required. The word's origin is essence, something with an odor or smell. Thus, the essential oils are those oils that have a fragrance, and the Essential Amphetamines are those compounds that can, in principle, be made from them by the addition of ammonia in the body.
There were a few interesting experimental trials that were based on these natural oils. Methoxyeugenol was assayed up to a 10 milligram level, and asarone at up to a 70 milligram level, and neither had any effects at all. And, in an attempt to challenge the "oil-to-amphetamine" concept, I made up a mixture of 1 part MDA, 2 parts TMA and 5 parts MMDA. A total of 100 milligrams of this combination (which I had named the "Pseunut Cocktail" for pseudo-nutmeg) should be equivalent to the safrole, elemicin and myristicin that would be in 5 grams of nutmeg. And 100 milligrams indeed produced quite a sparkle and considerable eye-dilation. But then, I have never taken 5 grams of nutmeg, so I cannot make any comparisons.
| [ |
[Main Index] | [Forward |

